Histopathologic Insights and Treatment Outcomes in PD-1 and PDL-1 Cutaneous Immune-Related Adverse Events: A Case Series
DOI:
https://doi.org/10.5195/ijms.2026.3588Keywords:
apremilast, anti-PD-1, anti-PDL-1, dupilumab, immune checkpoint inhibitors, lichenoid eruptions, psoriasiform rash, pembrolizumab, atezolizumab, case reportAbstract
Background: Immune checkpoint inhibitors (ICIs), including therapies targeting anti-programmed cell death protein 1 (PD-1) or anti-programmed death-ligand 1 (PDL-1), are highly effective for treating various malignancies, but are often associated with immune-related adverse events (irAEs). Among these, cutaneous irAEs are the most prevalent, affecting about half of patients and varying widely in severity. irAEs can impact quality of life and lead to treatment discontinuation. Managing these side effects effectively is essential to allow continuation of therapy without compromising its efficacy.
Methods: Retrospective case series.
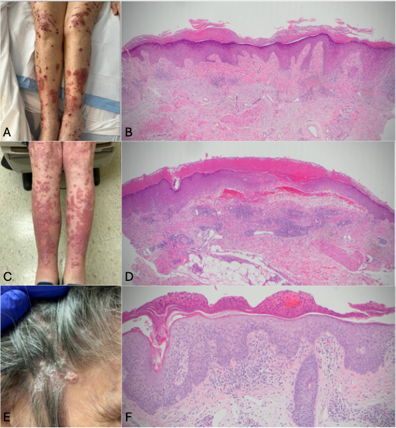
Results: We present three patients who developed severe cutaneous irAEs: two with pembrolizumab-induced lichenoid dermatitis and one with atezolizumab-induced psoriasiform rash. Initial treatment was guided by histopathologic findings, leading to the use of dupilumab, an interleukin-4 receptor (IL-4Ra) monoclonal antibody, in all three cases. While two patients achieved full resolution with dupilumab, the third case, which progressed to a clinically psoriasiform morphology, was later treated with apremilast, a phosphodiesterase 4 (PDE4) inhibitor, resulting in significant improvement.
Conclusion: These cases highlight the critical role of combining histopathologic and clinical insights to customize treatment approaches. Both dupilumab and apremilast are steroid-sparing options with favorable safety profiles and offer effective alternatives to systemic corticosteroids without compromising the efficacy of ICIs.
References
1. Quach HT, Johnson DB, LeBoeuf NR, Zwerner JP, Dewan AK. Cutaneous adverse events caused by immune checkpoint inhibitors. J Am Acad Dermatol 2021;85(4):956–966
2. Apalla Z, Papageorgiou C, Lallas A, Delli F, Fotiadou C, Kemanetzi C, et al. Cutaneous Adverse Events of Immune Checkpoint Inhibitors: A Literature Review. Dermatol Pract Concept 2021;11(1):e2021155
3. Phillips GS, Wu J, Hellmann MD, Postow MA, Rizvi NA, Freites-Martinez A, et al. Treatment Outcomes of Immune-Related Cutaneous Adverse Events. J Clin Oncol 2019;37(30):2746–2758
4. Zhang S, Tang K, Wan G, Nguyen N, Lu C, Ugwu-Dike P, et al. Cutaneous immune-related adverse events are associated with longer overall survival in advanced cancer patients on immune checkpoint inhibitors: A multi-institutional cohort study. J Am Acad Dermatol 2023;88(5):1024
5. Geisler AN, Phillips GS, Barrios DM, Wu J, Leung DYM, Moy AP, et al. Immune checkpoint inhibitor—related dermatologic adverse events. J Am Acad Dermatol 2020;83(5):1255
6. Sibaud V, Meyer N, Lamant L, Vigarios E, Mazieres J, Pierre Delord J. Dermatologic complications of anti-PD-1/PD-L1 immune checkpoint antibodies. Curr Opin Oncol 2016;28(4):254–263
7. Park JJ, Park E, Damsky WE, Vesely MD. Pembrolizumab-induced lichenoid dermatitis treated with dupilumab. JAAD Case Reports 2023;37:13
8. Kuo AM-S, Gu S, Stoll J, Moy AP, Dusza SW, Gordon A, et al. Management of immune-related cutaneous adverse events with dupilumab. J Immunother Cancer 2023;11(6):e007324
9. Mayor Ibarguren A, Enrique EA, Diana PL, Ana C, Pedro HP. Apremilast for immune checkpoint inhibitor-induced psoriasis: A case series. JAAD Case Rep 2021;11:84–89
10. Ellis S, Vierra AT, Millsop JW, Lacouture ME, Kiuru M. Dermatologic toxicities to immune checkpoint inhibitor therapy: A review of histopathologic features. J Am Acad Dermatol 2020;83(4):1130
Published
How to Cite
License
Copyright (c) 2026 Amanda Rodriguez Orengo, Alicia Mizes, Irina Lerman, Abigail Franco, Mary Gail Mercurio, Paul Blackcloud

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with this journal agree to the following terms:
- The Author retains copyright in the Work, where the term “Work” shall include all digital objects that may result in subsequent electronic publication or distribution.
- Upon acceptance of the Work, the author shall grant to the Publisher the right of first publication of the Work.
- The Author shall grant to the Publisher and its agents the nonexclusive perpetual right and license to publish, archive, and make accessible the Work in whole or in part in all forms of media now or hereafter known under a Creative Commons Attribution 4.0 International License or its equivalent, which, for the avoidance of doubt, allows others to copy, distribute, and transmit the Work under the following conditions:
- Attribution—other users must attribute the Work in the manner specified by the author as indicated on the journal Web site; with the understanding that the above condition can be waived with permission from the Author and that where the Work or any of its elements is in the public domain under applicable law, that status is in no way affected by the license.
- The Author is able to enter into separate, additional contractual arrangements for the nonexclusive distribution of the journal's published version of the Work (e.g., post it to an institutional repository or publish it in a book), as long as there is provided in the document an acknowledgment of its initial publication in this journal.
- Authors are permitted and encouraged to post online a prepublication manuscript (but not the Publisher’s final formatted PDF version of the Work) in institutional repositories or on their Websites prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work. Any such posting made before acceptance and publication of the Work shall be updated upon publication to include a reference to the Publisher-assigned DOI (Digital Object Identifier) and a link to the online abstract for the final published Work in the Journal.
- Upon Publisher’s request, the Author agrees to furnish promptly to Publisher, at the Author’s own expense, written evidence of the permissions, licenses, and consents for use of third-party material included within the Work, except as determined by Publisher to be covered by the principles of Fair Use.
- The Author represents and warrants that:
- the Work is the Author’s original work;
- the Author has not transferred, and will not transfer, exclusive rights in the Work to any third party;
- the Work is not pending review or under consideration by another publisher;
- the Work has not previously been published;
- the Work contains no misrepresentation or infringement of the Work or property of other authors or third parties; and
- the Work contains no libel, invasion of privacy, or other unlawful matter.
- The Author agrees to indemnify and hold Publisher harmless from the Author’s breach of the representations and warranties contained in Paragraph 6 above, as well as any claim or proceeding relating to Publisher’s use and publication of any content contained in the Work, including third-party content.
Enforcement of copyright
The IJMS takes the protection of copyright very seriously.
If the IJMS discovers that you have used its copyright materials in contravention of the license above, the IJMS may bring legal proceedings against you seeking reparation and an injunction to stop you using those materials. You could also be ordered to pay legal costs.
If you become aware of any use of the IJMS' copyright materials that contravenes or may contravene the license above, please report this by email to contact@ijms.org
Infringing material
If you become aware of any material on the website that you believe infringes your or any other person's copyright, please report this by email to contact@ijms.org







